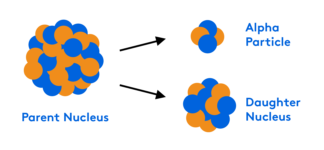
This webinar on salts and buffers is the second part in our three-part series on acid/base chemistry for the MCAT. You will learn about the three different types of salts, how to identify a buffer system, and how to calculate and predict pH depending on the buffer used. We will focus on the buffer system of the blood.
Salts and Buffers