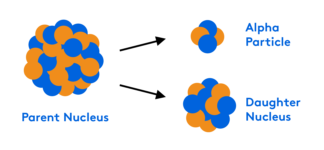
This webinar is the second part of our two-part series on acid/base chemistry. We will reinforce the salt and buffer concepts introduced in the prior webinar, how to recognize and successfully answer titration questions, and review the three different types of titrations. We will also cover how to read titration graphs and perform calculations to determine the concentration and identity of an analyte.
Acids and Bases, Part 2